EXPERIMENT OVERVIEW
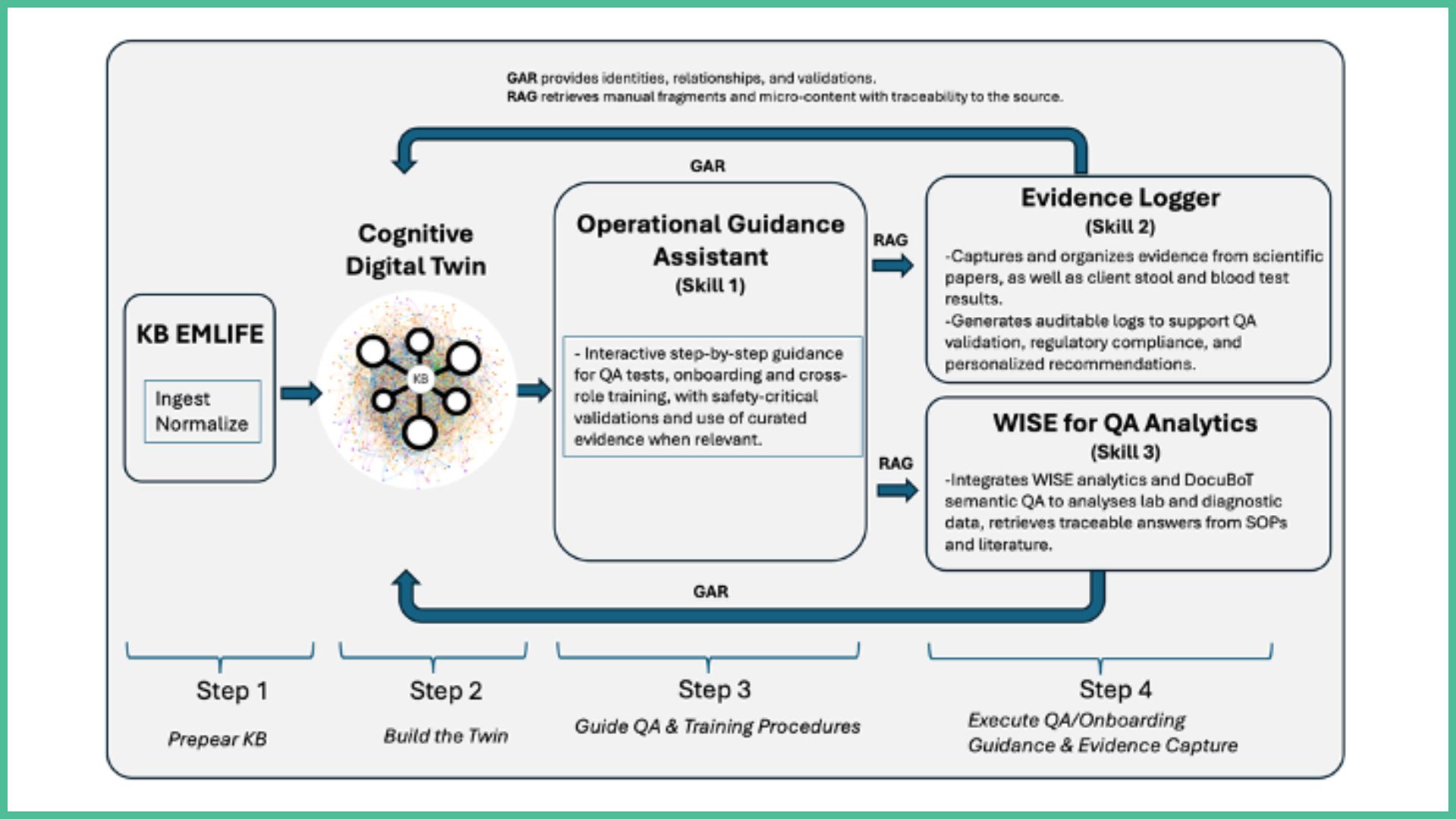
The proposed experiment will focus on the nutraceutical manufacturing sector, specifically the production of liquid probiotics at EMLIFE Biotics. This sector is characterized by complex fermentation processes, strict quality assurance requirements, and the need for highly trained operators. A key challenge is that much of the operational knowledge is fragmented: while around 20% of processes are already digitalized, another 60% remain in manual records, and approximately 20% are tacit knowledge not formally captured. The experiment aims to consolidate this expertise into a digital knowledge base and make it consumable through a Digital Intelligent Assistant (DIA) that supports employees across different stages of laboratory and production work.
The processes and tasks supported by the Digital Intelligent Assistant include quality assurance activities, where laboratory operators receive guidance during microbiological and physicochemical testing, are reminded of critical steps, and are supported in reducing the risk of errors or omissions. The assistant also supports the onboarding of new employees by guiding new staff step by step through laboratory and production protocols, shortening the learning curve and ensuring consistent adherence to safety and quality standards. In addition, it facilitates cross-role training by supporting logistics and packaging workers (“Type A”) in transitioning to production-related roles (“Type B”), providing interactive guidance and contextual operational knowledge.
The DIA will act as a supportive tool, not a replacement, empowering workers by making knowledge accessible in real time. For example, while formulations will remain based on standardized protocols, the assistant will provide contextual advice, highlight deviations, and suggest improvements, leaving final decisions to the human operator. By digitalizing and structuring EMLIFE’s production knowledge, the experiment will make it easier for
employees to access, apply, and share information in their daily work. This will strengthen quality control and reduce variability across production, while also accelerating the onboarding and reskilling of new staff. At the same time, the project safeguards tacit knowledge that could otherwise be lost, increasing the resilience of operations. In the long term, it will showcase how human-AI collaboration can enhance manufacturing, turning laboratory and production roles into positions that are more attractive, efficient, and sustainable.
EMLIFE operates in a nutraceutical manufacturing environment where production, laboratory testing, and quality assurance rely on a combination of documented procedures, manual records, and tacit knowledge accumulated through experience. While some processes are supported by Standard Operating Procedures (SOPs) and digital tools, a significant part of daily operations still depends on individual expertise, informal guidance, and fragmented information sources. This creates variability in execution and makes it difficult to ensure consistent knowledge transfer across teams.
One of the main challenges is related to onboarding and workforce development. Training new employees requires intensive supervision by experienced staff, and the quality of onboarding may vary depending on who delivers it. As a result, the learning curve is long, productivity is reduced during training periods, and the risk of procedural errors is higher. In parallel, internal mobility between packaging, production, and laboratory roles is limited due to the complexity of processes and the absence of structured, guided learning pathways.
Quality assurance and traceability represent another critical challenge. The production of live probiotic formulations requires strict compliance with Good Manufacturing Practice (GMP) standards and Hazard Analysis and Critical Control Points (HACCP) food safety systems, as well as accurate documentation of microbiological and physicochemical tests. In practice, quality-related information is distributed across different systems, paper records, and personal knowledge, which increases the effort required to retrieve, validate, and reconcile data. This can affect audit readiness, traceability, and overall operational efficiency.
From a technical and data perspective, EMLIFE faces difficulties in consolidating heterogeneous information sources into a unified and reusable framework. Process parameters, scientific references, quality records, and inventory data are not fully integrated, limiting their use for real-time decision support. This fragmentation reduces the potential value of existing data and prevents systematic learning from past production batches.
These challenges directly affect competitiveness, scalability, and operational resilience. As the company grows, dependence on key individuals, manual processes, and fragmented knowledge increases business risk and limits the ability to adapt quickly to market and production demands. Addressing these issues through a structured, human-centric Digital Intelligent Assistant (DIA) is therefore essential to improve reliability, workforce sustainability, and long-term industrial performance.
Objective 1:
To build a structured and validated digital knowledge base that consolidates EMLIFE’s laboratory and production procedures, scientific references, and quality requirements into a single cognitive digital twin, making critical operational knowledge accessible, traceable, and reusable across teams.
Objective 2:
To develop and deploy a Digital Intelligent Assistant that provides real-time, context-aware guidance to operators during quality assurance, onboarding, and daily production activities, while preserving human responsibility and ensuring compliance with regulatory and safety standards.
Objective 3:
To validate the assistant in real production conditions and demonstrate its impact on workforce development, process reliability, and internal mobility, including the facilitation of cross-role transitions from packaging to production roles without disrupting plant operations.
The PROBIO-AI experiment is carried out in the nutraceutical biotechnology sector, specifically in the manufacturing of liquid probiotic formulations based on live microbial cultures. The experiment takes place at EMLIFE’s production and laboratory facilities in Spain, within a real operational environment that includes fermentation processes, microbiological and physicochemical testing, bottling, and quality assurance activities. The project is implemented directly in day-to-day operations rather than in a simulated or isolated pilot setting, ensuring that results reflect practical industrial conditions.
The operational environment combines laboratory and production workflows, where strict coordination is required between testing activities, process monitoring, and quality validation. The assistant is deployed to support operators during routine and non-routine tasks, including quality checks, onboarding procedures, and cross-role training. Validation activities are conducted using real equipment, real production batches, and existing operational systems, allowing the experiment to assess usability and reliability under normal workload and time constraints.
The primary target users are laboratory technicians, production operators, and newly onboarded employees involved in fermentation, testing, and quality control. Secondary users include production supervisors and quality managers responsible for process validation and compliance. Relevant stakeholders also include technology providers, the Digital Innovation Hub, regulatory bodies, and professional partners who rely on EMLIFE’s compliance with quality and safety standards.
The experiment operates under established regulatory and operational constraints, including compliance with Good Manufacturing Practice (GMP) standards and Hazard Analysis and Critical Control Points (HACCP) food safety systems. Access to operational and quality data is governed by internal policies and data protection regulations, including pseudonymisation and controlled access rights. Integration with existing documentation systems, quality records, and inventory tools is required to ensure continuity of operations and avoid disruption of certified processes. These constraints shape the technical design of the assistant and ensure that innovation is compatible with certified industrial environments.
EXPECTED IMPACT
EXPECTED IMPACT
The PROBIO-AI experiment is expected to generate measurable operational, organisational, and strategic impacts for EMLIFE and its wider ecosystem. For end users, including laboratory technicians and production operators, the assistant will improve daily work conditions by providing reliable, contextual guidance during quality assurance, onboarding, and production activities. This is expected to reduce uncertainty, lower cognitive workload, and increase confidence when performing complex or safety-critical procedures. Success will be reflected in higher user adoption rates, reduced dependency on informal supervision, and faster attainment of operational autonomy by new employees.
From an operational perspective, the experiment is expected to improve process reliability and product quality by reducing information-related deviations, omissions, and inconsistencies. Targets include a reduction of at least 50% in errors linked to outdated or incomplete process information, a minimum of 95% traceability coverage across batches, and an improvement in inventory visibility and accuracy above 95%. These outcomes will be monitored through quality records, audit reports, and system usage logs.
Workforce development represents another key area of impact. By enabling structured onboarding and guided cross-role training, the experiment aims to reduce onboarding time by 30–40% and facilitate the transition of approximately 30% of packaging staff into production-related roles without disrupting plant operations. This contributes to greater organisational resilience and more sustainable workforce management.
In economic terms, improved efficiency, reduced rework, and lower training overheads are expected to contribute to cost savings and enhanced competitiveness. These benefits will be reflected in increased employee productivity, with a target improvement of 30–40%, and in shorter response times to audit and compliance requests, with an expected reduction of at least 70%.
The experiment also supports environmental and resource efficiency objectives. By reducing process deviations, material waste, and unnecessary repetitions of tests or batches, PROBIO-AI contributes indirectly to lower resource consumption and improved sustainability performance within the production process.
At ecosystem level, dissemination and visibility activities will ensure that project results reach relevant industrial and professional communities. A first impact target is the completion of the WASABI dissemination package by Month 12, including an experiment summary, documented lessons learned, and links to the published skill, supported by URLs and submission confirmations. In parallel, the project aims to achieve publication readiness in the WASABI White Label Shop by Month 12, demonstrated through official listings, acceptance messages, or screenshots.
The consortium will also produce at least one open-access communication asset, such as a public blog post, webinar, or short educational video, linked to the project’s dissemination plan and published with verifiable dates and links. In addition, at least one structured professional outreach action will be carried out towards relevant food, biotechnology, and healthcare communities, supported by agendas, invitations, or communication records.
Project visibility will be reinforced through milestone-based communication on LinkedIn, with four posts published at key stages: project start (Month 1), technical overview (Month 3), progress update (Month 9), and final results communication (Month 12). These publications will be documented through post URLs, screenshots, and publication dates, ensuring traceability and alignment with the overall communication strategy.
Overall, the success of PROBIO-AI will be assessed through a combination of technical performance indicators, workforce-related metrics, quality and compliance outcomes, and dissemination results. This integrated approach ensures that the experiment delivers measurable, sustainable, and transferable value for EMLIFE, the WASABI ecosystem, and other manufacturing SMEs.
GALLERY